
Thus,Įlectron pairs will spread themselves as far from each other as Whether or not they are in bond pairs or in lone pairs. The VSPER Theory states that electron pairs repel each other 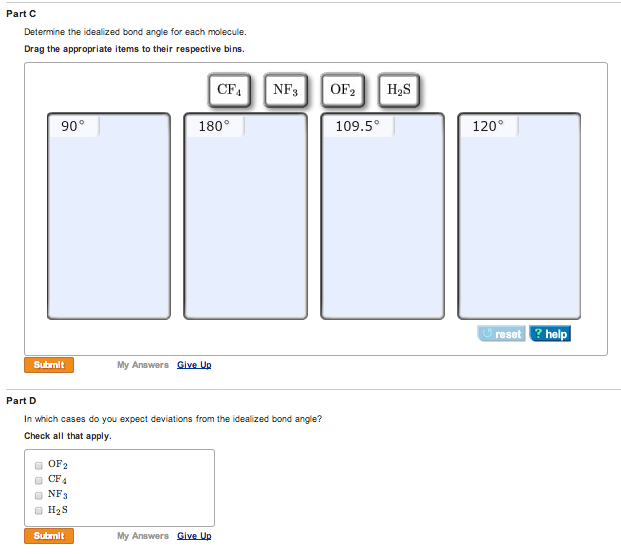
VALENCE SHELL ELECTRON PAIR REPULSION THEORY Lewis Structure we apply the Valence – shell electron pair repulsion (VSEPR)ĭraw the LEWIS ELECTRON DOT STRUCTURE of the following molecules. LEDS helps us to identify the bond pairs and the lone pairs. LEDS play a crucial role in determining the geometry of molecules because it Whether the sample is a solid, liquid, gas, or part of a solution. The molecular geometry of a substance may be different depending on Increasing the temperature gives the molecules more energy, which can lead to The best determination of a structure is made at low temperature because Infrared (IR) spectroscopy, Raman spectroscopy,Įlectron diffraction, and microwave spectroscopy. Molecules and learn about their vibrational andĮxamples include x-ray crystallography, neutron diffraction, Several analytical methods can be used to image MAED – SCIENCE / RAMON MAGSAYSAY TECHNOLOGICAL UNIVERSITYĪlso known as molecular structure, is a three dimensional Half-Life from Model Lake : 89.1 hours (3.CHEMICAL BONDING II: Molecular geometry and Hybridization of Atomic orbitals Removal In Wastewater Treatment (recommended maximum 95%): Half-Life from Model Lake : 89.1 hours (3.713 days) Half-Life from Model River: 0.9574 hours (57.44 min) Henry LC: 5.15 atm-m3/mole (Henry experimental database) Log BCF from regression-based method = 0.209 (BCF = 1.617) Rate constants can NOT be estimated for this structure!īioaccumulation Estimates from Log Kow (BCFWIN v2.17): Soil Adsorption Coefficient (PCKOCWIN v1.66):Īqueous Base/Acid-Catalyzed Hydrolysis (25 deg C) : Note: the sorbed fraction may be resistant to atmospheric oxidation OVERALL OH Rate Constant = 0.0000 E-12 cm3/molecule-secįraction sorbed to airborne particulates (phi): 7.46E-012 (Junge,Mackay) (m3/ug)):įraction sorbed to airborne particulates (phi):Ītmospheric Oxidation (25 deg C) : Structure incompatible with current estimation method! Hydrocarbon Biodegradation (BioHCwin v1.01): 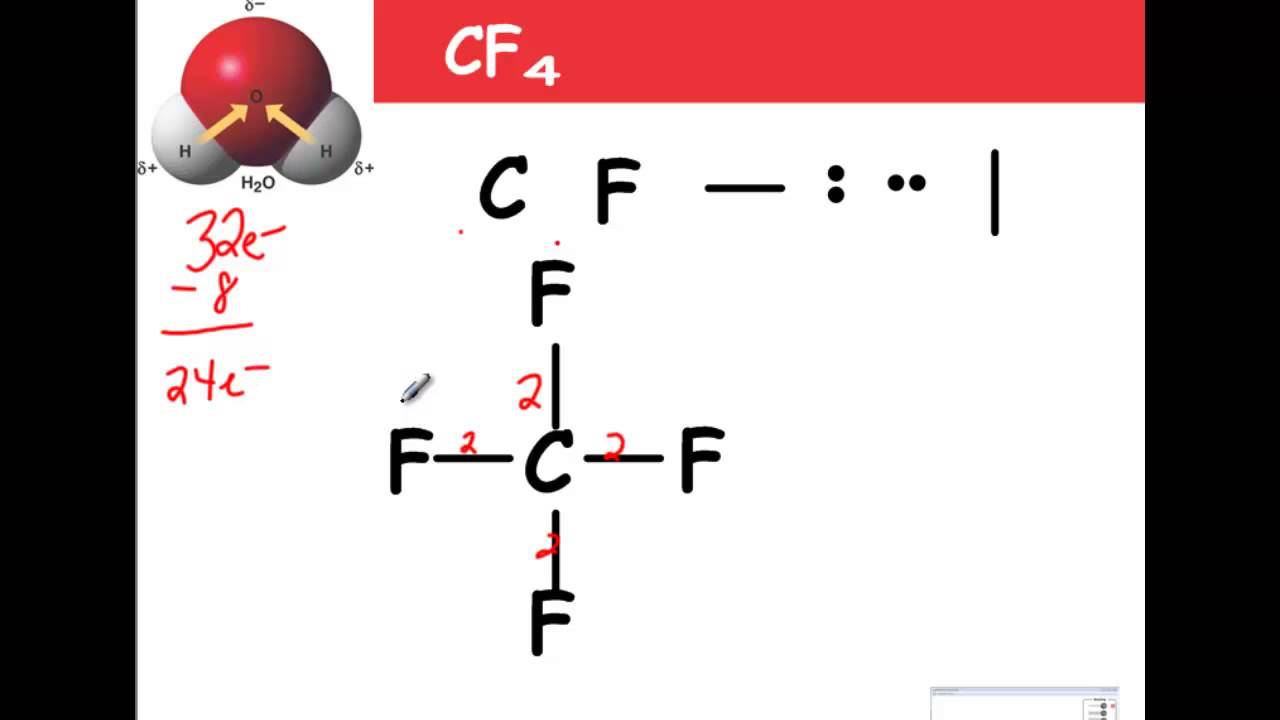
Probability of Rapid Biodegradation (BIOWIN v4.10):īiowin3 (Ultimate Survey Model): 2.7926 (weeks )īiowin4 (Primary Survey Model) : 3.5673 (days-weeks ) Log Octanol-Air Partition Coefficient (25 deg C) : Water Solubility at 25 deg C (mg/L): 2239 Water Solubility Estimate from Log Kow (WSKOW v1.41): VP (exp database): 1.75E+05 mm Hg at 25 deg C VP(mm Hg,25 deg C): 6.97E+004 (Mean VP of Antoine & Grain methods) Melting Pt (deg C): -164.82 (Mean or Weighted MP) Predicted data is generated using the US Environmental Protection Agency�s EPISuiteīoiling Pt, Melting Pt, Vapor Pressure Estimations (MPBPWIN v1.42):īoiling Pt (deg C): -83.29 (Adapted Stein & Brown method) Konukhova, S.V., Gas Chromatographic Identification of Ecologically Safe Freones, Vestn. (show more) ass: Semi-standard non-polar Column length: 2 m Column type: Packed Heat rate: 5 K/min Start T: 50 C End T: 220 C End time: 0 min Start time: 0 min CAS no: 75730 Active phase: Porapack Q Carrier gas: Nitrogen Data type: Normal alkane RI Authors: Zenkevich, I.G. Rodin, A.A., Gas chromatographic identification of some volatile toxic fluorine containing compounds by precalculated retention indices, J. (show more) ass: Semi-standard non-polar Column type: Capillary CAS no: 75730 Active phase: Porapack Q Data type: Normal alkane RI Authors: Zenkevich, I.G. Retention Index (Normal Alkane): 82 (Program type: Ramp Column cl.OU Chemical Safety Data (No longer updated) More details Safety: COMPRESSED GAS Matrix Scientific 006350Įnsure adequate ventilation.Incompatible with zinc, alkaline earth metals, Group I metals, aluminium and its alloys. Appearance: colourless gas OU Chemical Safety Data (No longer updated) More details.Experimental Density: -80 g/mL SynQuest.Experimental Vapor Pressure: 542 mmHg SynQuest.Experimental Boiling Point: -130 ☌ OU Chemical Safety Data (No longer updated) More details.183.6 ☌ Jean-Claude Bradley Open Melting Point Dataset 21272 184 ☌ Jean-Claude Bradley Open Melting Point Dataset 16142 184 ☌ OU Chemical Safety Data (No longer updated) More details Experimental Melting Point: -183.6 ☌ SynQuest.Experimental Physico-chemical Properties.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
